Dry ice and millions of secret chickens
When the government absolutely has to get involved in production
This week I’ve been thinking a lot about all of the markets that have to function for the COVID-19 vaccine distribution to take place. There’s almost too many to name—vials, syringes, dry ice, cargo flights, saline, gloves.
I’ve also been wondering why we weren’t more prepared. Pandemics of the COVID-19 magnitude are rare, but we experience an almost-pandemic or a milder pandemic every few years. The flu season of 2014-2015 was particularly bad, as was H1N1 in 2009-2010.
The answers, from what I’ve learned, are complicated:
First, the government does prepare for pandemics. A few years after the anthrax scare of 2001, the government formed BARDA (Biomedical Advanced Research and Development Authority), an agency under the Department of Health and Human Services. The role of BARDA is to prepare against biological threats, including biochemical weapons and the inevitable pandemic.
But we were not prepared for COVID-19 for several reasons.
After the SARS outbreak ended, research into coronaviruses became less popular. Not only did it seem like coronaviruses were of low importance, but research on a potential SARS vaccine ground to a halt. Why? Because the way we currently run vaccine trials requires the virus to be actively circulating in the population.
🦠 Bioethics side note: after many years (centuries) of scientists using vulnerable populations as testing grounds for vaccines (in what we now recognize as a horrible abuse of power), the 1978 Belmont Report created some firm ethical lines (that most scientists today take very, very seriously).
The ethical principles outlined in the Belmont Report created some difficulties during the COVID-19 vaccine trials—most scientists are very uncomfortable with the idea of deliberately infecting even willing, young volunteers with a virus as unknown as COVID-19.
So instead of conducting so-called challenge trials (where volunteers would be purposely infected post-vaccination to see if the vaccine works), all of the COVID-19 vaccine trials to date have had to wait for their vaccinated group and their control group to become exposed to the virus in the course of their daily lives. Which requires a virus to be actively circulating in the population. This is part of the reason for the delay between the first injections and the final results.
Ironically, the COVID-19 vaccine trials didn’t meet the level of viral exposure they needed to complete the study until the fall COVID-19 wave in the U.S.
We expected the next pandemic to be caused by the influenza virus. (There’s a complicated history here involving our close proximity to birds and the 1918 flu pandemic, but I’ll skip it.)
The pharmaceutical market is highly concentrated and calcified; the pursuit of science for science’s sake has largely fallen away in pursuit of forever growing shareholder value, and so we’re less prepared to quickly pivot into new areas. These factors are magnified with the vaccine market; because pandemics require a large upfront investment with no guarantee of return, the federal government usually has to motivate (fund) forays into vaccine research.
The global markets that handle commodities that go into fighting pandemics (the aforementioned syringes, vials, gloves, dry ice, etc.) are hinged on the just-in-time thesis. Just-in-time, or JIT, was pioneered in Japan as a way to save money by keeping just the bare minimum of inventory in stock. For example, Toyota stocking just enough parts for one day at a time of car production. Using a JIT supply chain means there’s less money tied up in inventory, and the company runs leaner and has more cash around. But JIT also makes supply chains much more prone to disruption.
Given that background, I want to talk about two things. First, how we might face a dry ice shortage during the initial phase of the COVID-19 vaccine roll out. Second, how the federal government has stepped in in the past to rectify the market distortions caused by a pandemic during an era of global JIT supply chains, using chickens as an example.
Dry ice
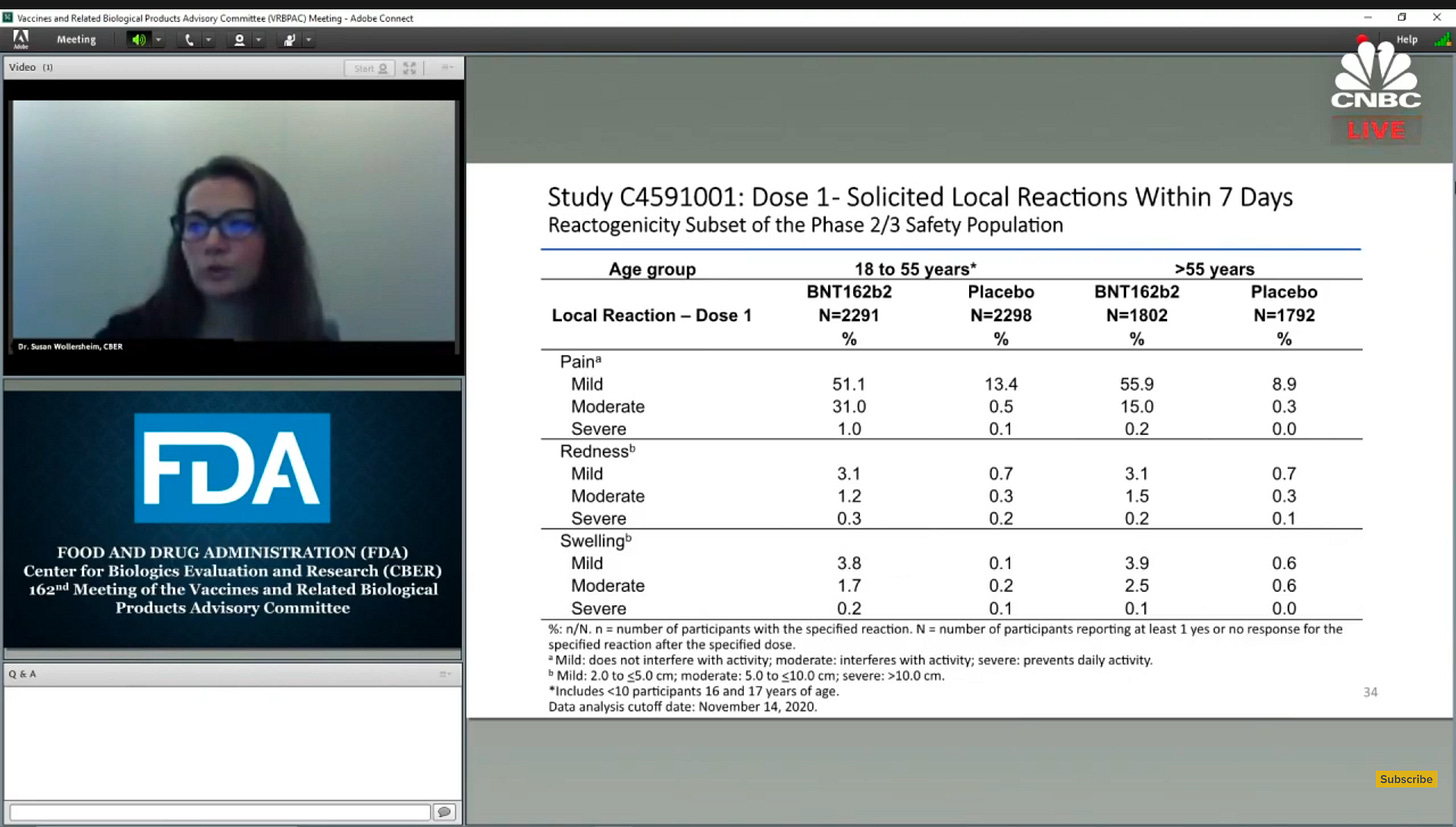
The Pfizer vaccine, likely to be approved by the FDA today or tomorrow, will be shipping across the country on dry ice. Will this cause shortages? It looks like it might.
But I started to wonder, after the hosts of the This Week in Virology podcast brushed aside concerns about a shortage (note: TWiV hosts, I love you, lmk if you ever come out with a branded mug). After all, dry ice is just a solid form of carbon dioxide, the gas we exhale.
However, the simplicity of a product doesn’t necessarily correspond to whether there will be a shortage. After Hurricane Maria hit Puerto Rico in 2017, shutting down several medical saline manufacturing plants, there was a national shortage of saltwater. If there are only a few global suppliers of a product, it doesn’t matter how simple it is, there will be shortages if the market is disrupted by national disaster or the demand of a pandemic.
This looks to be the state of the dry ice market. According to Thomasnet.com, an online platform for product sourcing, there are just a few major producers of dry ice in the U.S.
Source: https://www.thomasnet.com/articles/top-suppliers/dry-ice-suppliers-manufacturers/
These producers are used to making a set amount of dry ice every year, and they may very well struggle to meet the increased demand of the pandemic. The dry ice shortage is likely to be exacerbated by the pandemic; apparently the carbon dioxide used to produce dry ice is typically captured as a byproduct of ethanol processing for fuel. And because Americans are using far less fuel during the pandemic, the rate of ethanol processing has gone down. (A side note: I haven’t been able to figure out if medical dry ice production has different suppliers or sources—sometimes the medical industry requires an additional layer of sterility, but I’m not sure if that applies here.)
The American supply of secret chickens
Listeners of the NPR Planet Money podcast will be familiar with the story I’m about to tell, which covers how the federal government, frightened by the possibility of a flu pandemic, decided to shore up the weaknesses of a certain market: chicken eggs.
Most influenza in humans comes from birds. So when vaccine manufacturers need to grow a lot of virus to make flu vaccines, they grow them in chicken eggs. The virus is then killed and the egg white is washed off before it is packaged as a final vaccine. (The science around making flu vaccines is changing, but currently about 90-95% of our flu vaccines are made using flu viruses grown in chicken eggs.)
Around 2004, when BARDA was formed because the government expected bioterrorism and recurrent pandemics to change the landscape, smart thinkers started to realize that the market for chicken eggs was just like that for dry ice. Chickens only lay one egg a day, and the American people only use so many eggs a year. No for-profit corporation wants to feed extra chickens only to potentially throw the eggs out. So the federal government decided to contract with several corporations for a supply of several million chicken eggs every year.
The details around these contracts are purposely vague—not even the name of the companies, the location of the farms, or the number of chicken eggs is publicly available—for national security purposes. But we do know that HHS has at least one contract for $42 million (as of 2017) with a corporation that sources its eggs in the U.S., and at least three other contracts with corporations that source their eggs globally.
In the years that no emergency flu vaccine is needed, the excess millions of eggs owned by the federal government are turned into animal feed. And in the event of a pandemic, the federal government has helped shore up the warped market that would catch us all unprepared.
Conclusion
The market for commodities is more rickety and prone to disruption than you’d expect, and the vaccine roll-out is going to strain it even further. Any single step of the roll-out could cause a shortage that delays the vaccination process.
But with the secret chicken stockpile, the government has one proven way to shore up these systems. To prevent future shortages in essential supplies, the government will have to step in a lot more.
Happy likely vaccine approval day!