Peptides, compounding, and a disdain for regulation
The FDA is finally cracking down on Hims
ICYMI, I started a second newsletter, where I’ll be writing more explicitly political/policy-focused pieces. The first one is coming out later this week with an exciting guest interview. You can subscribe here:
Peptide parties
If you don’t live in San Francisco, you might not have heard of the peptide craze. In fact, if you do live in SF, even mentioning it might feel out of date — peptide parties have been going on for months at least. But most of the country has not heard of this.
If you’re one of us who does not live in SF: Peptides are unlicensed, unproven drugs that are easily accessible from online vendors, and for which there have been peptide-injection parties. The idea is that there’s a peptide for everything; as GLP-1s are to obesity and weight loss, so are other peptides for insomnia, ADHD, rosacea, or any other ailment, real or imagined.
These peptides are mostly coming from vendors based in China, which may or may not be manufacturing the peptides themselves (many Chinese manufacturers — as do other manufacturers globally — subcontract out). They’re unlicensed and unregulated, so even if the FDA was good at touring overseas factories (it’s not), these would not fall under the FDA’s purview.
I’m not sure what’s happened with pharmaceuticals in American culture recently — it’s probably a combination of MAHA influence and a built-up sense that the FDA takes too long to approve new drugs and is overall too strict — but long-held shibboleths are changing. Dr. Marty Makary, head of the FDA, recently suggested that all drugs should be available over the counter unless they’re addictive or dangerous. There are these Chinese peptides. And compounding DTC companies are almost entirely out of control.
Almost.
Pushing the FDA…
Hims, one of the big DTC telehealth companies that prescribes medications, and which went public in a SPAC in 2021, has been on the vanguard of making pharmaceuticals accessible to patients outside the traditional bounds of what the FDA would allow.
Compounding these drugs, or taking active pharmaceutical ingredients and doing the last steps of drug manufacturing at a small scale at compounding pharmacies, is legal under certain circumstances, including if the drug meets certain conditions and is in shortage. GLP-1s were declared to be in shortage in 2022, which opened the door for telehealth companies like Hims to compound these drugs. They sold them for a fraction of the brand name price, directly competing with on-patent drugs, a move so bold as to be nearly unprecedented. Then, in April 2025, the FDA declared that GLP-1s were no longer in shortage.
Related articles
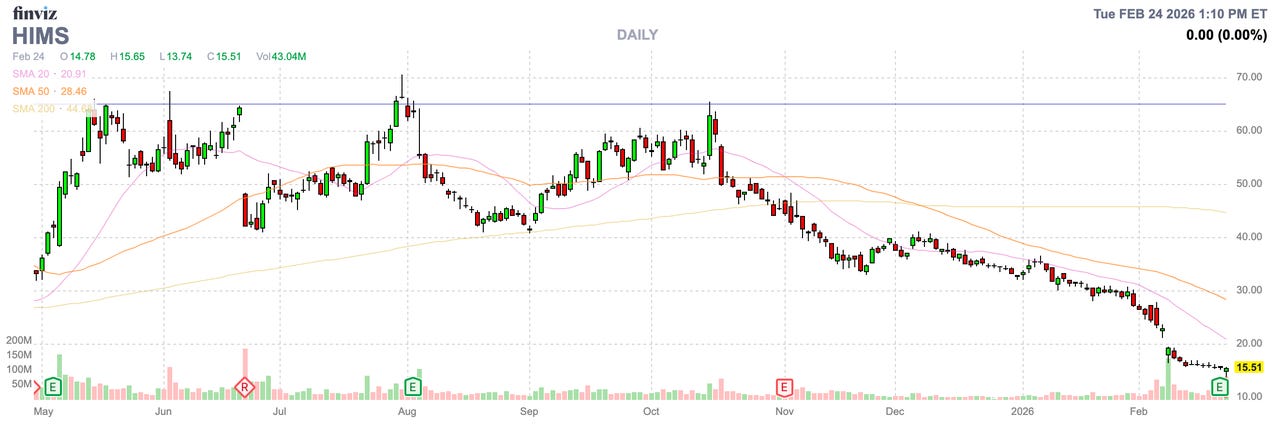
Novo Nordisk, the manufacturer of Wegovy and Ozempic, took an incremental approach to this, perhaps not believing that Hims would continue to flout the rules so blatantly. In June 2025, Novo announced that it would no longer partner with Hims. Although Hims’ stock price dropped on the announcement, the company’s CEO framed the Novo decision as anticompetitive and Hims continued to sell GLP-1s.
…past its limits
During the Super Bowl this year, Hims aired an ad that framed its compounded drugs as a way of democratizing access to the expensive lifespan-lengthening treatments that the wealthy are using. In its blog post on the ad, Hims noted that “[i]f our message makes the industry uncomfortable, it’s because their profits rely on the many but benefit the few.”
Then, Hims debuted a compounded version of Novo Nordisk’s semaglutide oral pill. Combined, these actions seem to have been a bridge too far for both the FDA and Novo Nordisk; the FDA named Hims in an announcement that it was taking “decisive steps” against compounded GLP-1s, and HHS asked the DOJ to investigate Hims. Novo Nordisk finally filed suit. Hims announced it would stop selling the compounded pill.
Now what?
Hims was making a bet that, as with the peptide parties, the general approach toward pharmaceutical regulation was relaxed under the current administration. It wasn’t a crazy bet. As I wrote above, Makary is interested in reforming key tenets of American pharmaceutical regulation, like keeping drugs behind the counter.
But Hims pushed it too far when it copied the semaglutide pill. As STAT’s Readout Loud podcast pointed out, compounding an oral GLP-1 is particularly difficult chemically, meaning that Hims was potentially introducing new risks to its customer base. And also if the FDA were to allow Hims to proceed unencumbered, it would upend decades of patent law and tradition around pharmaceuticals (not that that would necessarily be the worst thing, but it definitely shouldn’t happen haphazardly).
What will happen to Hims? The always-worthwhile Health Tech Nerds newsletter reviewed Hims’ earnings call last week:
[I]f you took away all identifying details and told me a company generating $2.3 billion of revenue, growing 59% YoY, with an Adjusted EBITDA margin of 14%, is only trading at an enterprise value around $3.8 billion, I’d tell you that seems like a great investment. Yet when you tell me that the company is Hims, and their management team neglected to mention on the earnings call that they’re under SEC investigation, after all the recent shenanigans, I very much see why investors are skeptical here.
I’m not a culture writer — but taken together, the general public reaction to peptides (so far a very localized phenomenon) and the compounded semaglutide seems to indicate that we’re ready for a loosening up of the pharma rules. Is that because the FDA has done such a great job over the last few decades, such that Americans can’t picture a drug being really harmful? Or is it because of a general desire for democratization of access, like Hims was trying to market?
But the bigger question is how (and if) the FDA manages to reform pharmaceutical regulation, while cracking down on companies like Hims that have gone too far, while maintaining its reputation as one of — if not the — finest pharmaceutical regulatory agencies in the world. It’s a tall order.


Great post. Peptides are getting hot here in miami too